Vascular Targeting
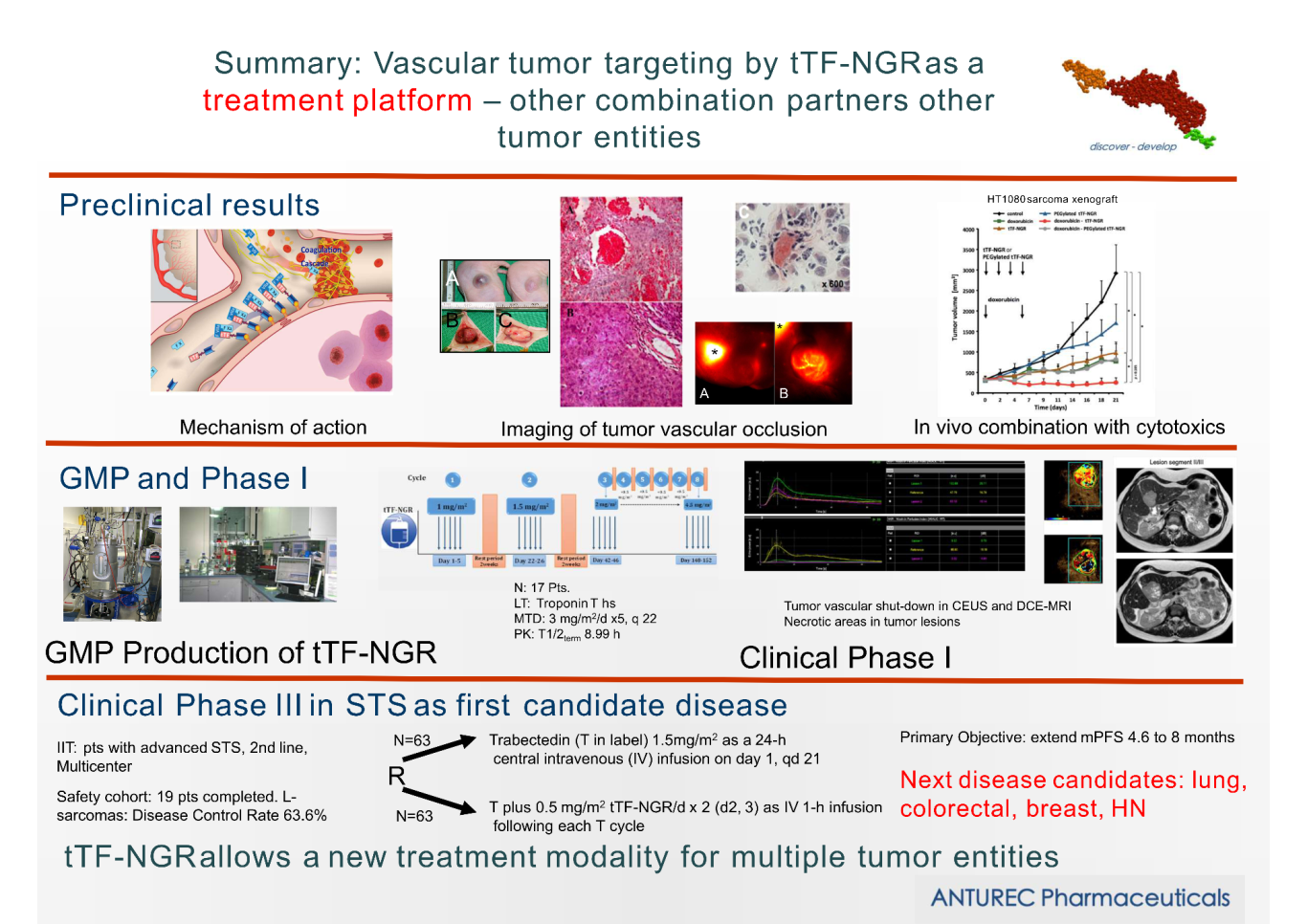
ANTUREC's mission embraces research and development in the area of new targeted recombinant drugs against cancer and the translation of promising compounds from the laboratory to the clinic. ANTURECs first ANti TUmor RECombinant pharmaceutical – the Investigational Medicinal Product (IMP) tTF-NGR – is in clinical phase III studies in oncology. In addition to the clinical lead structure tTF-NGR, ANTUREC follows active preclinical programs on other targeted proteins.
tTF-NGR: By targeting tissue factor, a central molecule within the extrinsic blood coagulation pathway, to tumor vasculature we aim to selectively occlude tumor vessels and induce tumor infarction. Our target is aminopeptidase N (CD13) located on cells of the tumor vasculature, such as invasive endothelial cells and pericytes and some tumor cells, but absent from most mature blood vessels. We have designed and studied truncated tissue factor (tTF) with a C-terminal GNGRAHA peptide (tTF-NGR) binding to CD13 and causing tumor vascular thrombosis with subsequent tumor infarction.
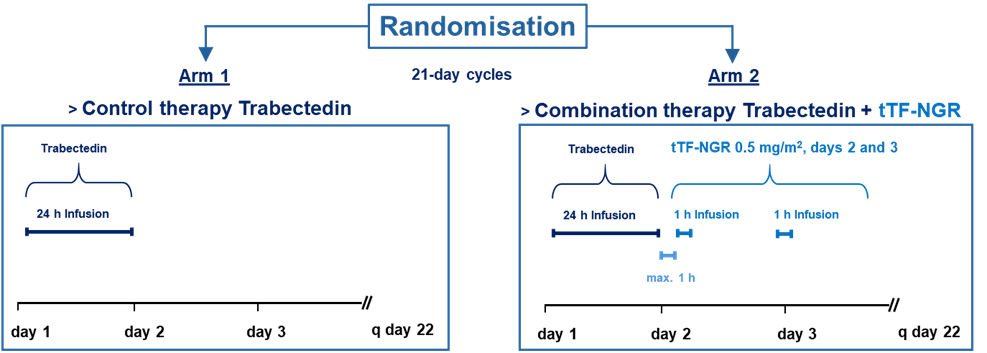
In our clinical studies we combine tTF-NGR with cytotoxic drugs. When the tTF-NGR is given at the time of high intratumoral cytotoxic drug levels, the tumor vessels will be occluded resulting in tumor-entrapped drugs. By this mechanism, the antitumor activity of cyctotoxic drugs can be significantly enhanced. Vice versa, the activity of tTF-NGR is increased by the apoptotic action of the cytotoxic drugs on tumor cells and tumor endothelial cells through upregulation of phosphatidylserine. This principle is tested in a first randomized clinical phase III study in patients with advanced soft tissue sarcoma (TRABTRAP, EU CT NUMBER 2024-516392-33-00).
As tTF-NGR targets the tumor vasculature, which is similar in different tumor types, the principle of entrapping tumor therapeutics by tTF-NGR induced vascular occlusion, if successful in the active trial, can be generalized as a treatment platform for multiple vascularized tumor entities.
Summarized schedule of the randomized phase of the clinical study TRABTRAP